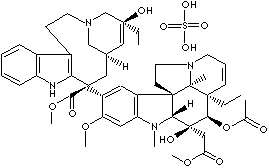
PRODUCT IDENTIFICATION
H.S. CODE
TOXICITY
PRICE
CLASSIFICATION
PHYSICAL AND CHEMICAL PROPERTIES
REFRACTIVE INDEX
NFPA RATINGS
Health: 2, Flammability: 1, Reactivity: 0
AUTOIGNITION
FLASH POINT
GENERAL DESCRIPTION & APPLICATIONS
Vinca alkaloids, including the natural products vincristine and vinblastine and the semisynthetic derivatives vindesine and vinorelbine, are antimitotic drugs that are widely used in the retreatment of cancer. Vinorelbine, a microtubule inhibitor, is the latest of the Vinca alkaloids, which promotes apoptosis in several tumor cell lines and in vitro shows both multidrug and non-multidrug resistance. Vinorelbine induce disassociation of cellular microtubules, most likely by modifying the dynamics of the spindle microtubules. This suppression of microtubule dynamics appears to cause a mitotic block by activating the metaphase-anaphase checkpoint. Vinorelbine arrests cell cycle and inhibits microtubule assembly by binding tubulin and inducing coiled spiral aggregate formation. Microtubules are present in mitotic spindles, neuronal axons, and other cells. Inhibition of mitotic microtubules appears to correlate with antitumor activity, while inhibition of axonal microtubules seems to correlate with neurotoxicity. Compared to vincristine and vinblastine, vinorelbine is more selective against mitotic than axonal microtubules in vitro, which may account for its decreased neurotoxicity. Like the other Vinca alkaloids, vinorelbine has a potency in the submicromolar range.(source: Sigma-Aldrich)
Mitotic inhibitors are often plant alkaloids and other compounds derived from natural products. They can stop mitosis or inhibit enzymes from making proteins needed for cell reproduction. These work during the M phase of the cell cycle but can damage cells in all phases. They are used to treat many different types of cancer including breast, lung, myelomas, lymphomas, and leukemias. These drugs are known for their potential to cause peripheral nerve damage, which can be a dose-limiting side effect. (source: American Cancer Society)
Mitotic Inhibitors
|
Product |
CAS RN. |
|
Vincristine |
57-22-7 |
| Colchicine | 64-86-8 |
| 2-Methoxyestradiol | 362-07-2 |
| Demecolcine | 477-30-5 |
| Podofilox | 518-28-5 |
| Vinblastine | 865-21-4 |
| Trifluralin | 1582-09-8 |
| Estramustine | 2998-57-4 |
| Benomyl | 17804-35-2 |
| Oryzalin | 19044-88-3 |
|
Teniposide |
29767-20-2 |
| Nocodazole | 31430-18-9 |
| Mebendazole | 31431-39-7 |
| Paclitaxel | 33069-62-4 |
| Etoposide | 33419-42-0 |
| Maytansine | 35846-53-8 |
| Vindesine | 53643-48-4 |
| Albendazole | 54965-21-8 |
| Phomopsin A | 64925-80-0 |
| Ansamitocins | 69279-90-9 |
| Vinorelbine | 71486-22-1 |
| Combretastatin | 82855-09-2 |
| Rhizoxin | 90996-54-6 |
| Dolastatin 10 | 110417-88-4 |
| Cryptophycin | 124689-65-2 |
| Disermolide | 127943-53-7 |
| Docetaxel | 148408-66-6 |
| Epothilone B | 152044-54-7 |
| Ixabepilone | 219989-84-1 |
APPEARANCE
ASSAY
96.0 - 102.0% (on dried basis)
ETHYL ALCOHOL
15.0% max
ACETONE
0.1% max
HEAVY METALS
20ppm max
15.0% max